Neurodegenerative Research
CHEMISTRY, BIOCHEMISTRY, PHARMACOLOGY, PROTEIN SURFACE MIMETICS, GPCR REGULATION, ENZYME REGULATION, ANTI-INFLAMMATORY RESEARCH, ANTI-INFECTIVES RESEARCH, ANTI-CANCER RESEARCH, NEURODEGENERATIVE RESEARCH, CARDIOVASCULAR RESEARCH
We are using our novel compounds discovered from antiinflammatory and amyloid research programs to penetrate the blood-brain barrier and regulate important physiological processes that become aberrant in neurodegenerative diseases.
1) Beta Amyloid Peptides and Proteins (Harrison et. al 2007)
2) Protease Inhibitors In The Brain
3) GPCR Regulation In The Brain
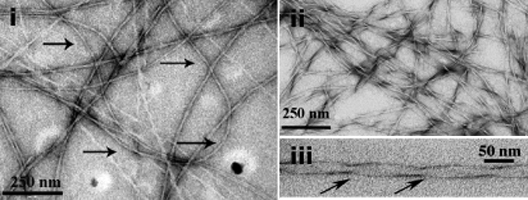
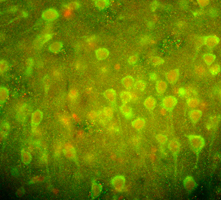
Figure: (Left): Amyloid peptide. i-iii : Typical electron micrographs of amyloid fibrils showing their linear, unbranched and flexible nature (i) and repeating twists of fibrils at regular intervals (ii & iii). (Far Right): A GPCR known as PAR2 (green) and one of its activating proteases, trypsin (red), coexpressed in neurons of the cerebral cortex of the rat (immuohistochemical photomicrograph).
PUBLICATIONS:
Alpha-synuclein structure and Parkinson's disease - lessons and emerging principles. Meade RM, Fairlie DP, Mason JM. Mol Neurodegener 2019,14, 29.
Protease activated receptor 2 controls myelin development, resiliency and repair. Yoon H, Radulovic M, Walters G, Paulsen AR, Drucker K, Starski P, Wu J, Fairlie DP, Scarisbrick IA. Glia 2017, 65, 2070-2086.
Toward peptide-based inhibitors as therapies for Parkinson's disease. Mason JM, Fairlie DP. Future Med Chem 2015, 7, 2103-2105.
C5a receptor (CD88) inhibition improves hypothermia-induced neuroprotection in an in vitro ischemic model. Thundyil J, Pavlovski D, Hsieh YH, Gelderblom M, Magnus T, Fairlie DP, Arumugam TV. Neuromolecular Med 2012, 14, 30-39.
Amyloid formation from an α-helix peptide bundle is seeded by 310-helix aggregates. Singh Y, Sharpe PC, Hoang HN, Lucke AJ, McDowall AW, Bottomley SP, Fairlie DP. Chemistry Eur J. 2011, 17, 151-160.
Amyloid peptides and proteins in review. Harrison RS, Sharpe PC, Singh Y, Fairlie DP. Rev Physiol Biochem Pharmacol. 2007, 159, 1-77. [>100 citations]
Inhibitors of beta-amyloid formation based on the beta-secretase cleavage site. Abbenante G, Kovacs DM, Leung DL, Craik DJ, Tanzi RE, Fairlie DP. Biochem Biophys Res Commun. 2000, 268, 133-135.
Is Oxidative Damage By Beta Amyloid and Prion Peptides Mediated by Hydrogen Atom Transfer from Glycine Alpha-Carbon to Methionine Sulfur within Beta-Sheets? A. Rauk, D. A. Armstrong, and D. P. Fairlie. J. Am. Chem. Soc. 2000, 122, 9761-9767.
Characterization of copper interactions with alzheimer amyloid beta peptides: identification of an attomolar-affinity copper binding site on amyloid beta1-42. Atwood CS, Scarpa RC, Huang X, Moir RD, Jones WD, Fairlie DP, Tanzi RE, Bush AI. J Neurochem. 2000, 75, 1219-1233. [>100 citations]
Solution structures in aqueous SDS micelles of two amyloid beta peptides of A beta(1-28) mutated at the alpha-secretase cleavage site (K16E, K16F). Poulsen SA, Watson AA, Fairlie DP, Craik DJ. J Struct Biol. 2000, 130, 142-152.
Cu(II) potentiation of alzheimer abeta neurotoxicity. Correlation with cell-free hydrogen peroxide production and metal reduction. Huang X, Cuajungco MP, Atwood CS, Hartshorn MA, Tyndall JD, Hanson GR, Stokes KC, Leopold M, Multhaup G, Goldstein LE, Scarpa RC, Saunders AJ, Lim J, Moir RD, Glabe C, Bowden EF, Masters CL, Fairlie DP, Tanzi RE, Bush AI. J Biol Chem. 1999, 274, 37111-6. [>100 citations]
Aqueous dissolution of Alzheimer's disease Abeta amyloid deposits by biometal depletion. Cherny RA, Legg JT, McLean CA, Fairlie DP, Huang X, Atwood CS, Beyreuther K, Tanzi RE, Masters CL, Bush AI. J Biol Chem. 1999, 274, 23223-23228. [>100 citations]
The possible origin of free radicals from amyloid beta peptides in Alzheimer's disease. Bush AI, Huang X, Fairlie DP. Neurobiol Aging 1999, 20, 335-337.
The Solution Structure of Amyloid β-Peptide (1-40) in a Water-Micelle Environment. Is the membrane spanning domain where we think it is? Coles, M.; Bicknell, W.; Watson, A. A.; Fairlie, D. P.; Craik, D. J. Biochemistry 1998, 37, 11064-11077. [>100 citations]
Solution structure of methionine-oxidized amyloid beta-peptide (1-40). Does oxidation affect conformational switching? Watson AA, Fairlie DP, Craik DJ. Biochemistry 1998, 37, 12700-12706. [>100 citations]

